i refuse to read that, what is the cliffs notes version?
Here's the quick and dirty version:
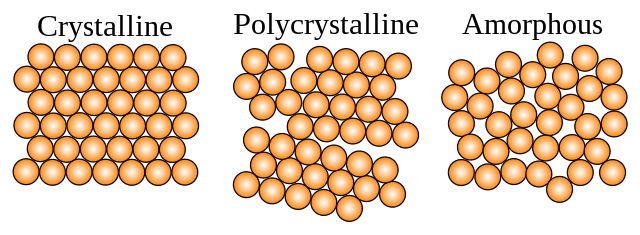
All solids exist in one of 3 possible flavors - Crystals, polycrystals and glasses.
Crystals are when atoms or molecules come together in a stacked order, kinda like a rack of billiards balls.
Polycrystals are the same thing, only now there are many stacks/racks facing in different directions (also known as grains).
Glasses are a jumbled mess, like inside a bag of Skittles. No order exists past the size of molecule.
Metals are normally poly-crystalline. This research paper focuses on the techniques used to discover more metallic combinations that will readily solidify into a glass. What they are doing is kind of 'brute-forcing' the discovery of a new and useful material: Discover a few glasses and
maybe a couple of them are useful for something. Get a computer to find hundreds more, and now things are looking promising!
Heat-treating glass works though different principles from other solids.
Grain size? Carbides? None'o dat in a glass... but we can still temper, anneal and harden it. Although you wouldn't want to drop a differentially hardened glass knife during a busy dinner service. Exploding glass would go
everywhere...
But really, it's like we're going back in time. Obsidian blade from the stone age? Totally a glass knife.
Hopefully we get some good things out of what these guys are doing with their research. Godspeed.
No choil shot?
Bonus round:
Crystalware was
really badly named. I mean, there are
no crystals in crystalware. Instead, it's the polar opposite; made entirely of glass.
C'mon guys, you had one job...